 Source: Stefano Lugli
Source: Stefano Lugli
The Arbiozome project, coordinated by the Pugère research station, was carried out over three consecutive years, from 2022 to 2024. It was based on the aggregation of experiments conducted at three sites, each monitored by a different partner, to assess the potential impact of biostimulants on tree crops. At the Tapy research station, four treatments were compared with a control treatment, all in cherry orchards but with a variable range of measurements.
In France, cherry production is strongly concentrated in three main agricultural regions: Provence-Alpes-Côte d’Azur (36%), Auvergne-Rhône-Alpes (33%) and Occitanie (22%). National production is estimated at around 32,000 tonnes per year (source: Annual Agricultural Statistics). Vaucluse is the leading department for cherry production, accounting for around 25% of the national tonnage of cherries for fresh consumption.
French fruit production, and cherry cultivation in particular, is in sharp decline. In the south-east, the cultivated area has fallen by 10–30% in ten years (Agreste, 2011), due to pressure on land use (urbanisation, infrastructure) and declining farm profitability. In addition, the arrival of difficult-to-control pests, such as Drosophila suzukii, has strongly discouraged growers, contributing to the reduction in cultivated area from 15,000 hectares in 1989 to 8,000 hectares in 2020.
Establishing an orchard represents a significant investment, up to €60,000, with insect-proof protection lasting 15 to 20 years. This transition is taking place in a context where mechanisation remains limited and labour requirements are very high, reaching 900 hours/ha/year. Added to this are agronomic challenges (soil depletion, compaction, root rot), phytosanitary issues (diseases, loss of authorised active substances) and social concerns (regulatory pressure, the image of the profession).
The gradual degradation of soils, caused by decades of monoculture without crop rotation, depletion of organic matter and compaction resulting from tractor traffic, represents the main challenge for the sector. This degradation slows the establishment of young trees, which now take five years to become established, compared with two or three years in the past. It causes yield losses of up to 50%, despite the implementation of soil improvement or replanting strategies. Fertilisation therefore remains a crucial tool, but its effectiveness is limited by the low bioavailability of nutrients, particularly phosphorus, which is often bound in the soil. Poor nitrogen management can lead to various forms of pollution (water pollution and greenhouse gas emissions). Effective fertility depends largely on soil biological activity, which is still widely overlooked.
Faced with these limitations, the sector is seeking to move towards more sustainable practices. This transition involves reducing inputs, improving soil microbial biodiversity and using alternative solutions such as biofertilisers based on so-called “effective” microorganisms. These could help restore natural balances, improve nutrient uptake and reduce environmental impact (carbon, nitrogen, phosphorus).
The project aims to test the use of biofertilisers in orchards under different conditions, in order to measure their impact on soils and crops. The objective is to provide benchmarks to support the sector in moving towards greater autonomy, sustainability and profitability, in an increasingly demanding economic, environmental and social context.
As part of efforts to improve agronomic practices, a study was conducted to assess the impact of biofertilisers in orchards. This study focused on three separate trials. The first concerned cherry trees and was carried out at the CTIFL research station in La Tapy. The other two trials involved apple trees: one at the La Pugère station and the other at the La Morinière station. This article presents the results obtained for cherry trees at the La Tapy research station.
The biofertilisers tested are commercially available solutions in France and are easily accessible to growers. These products are mainly designed to inoculate the soil with microorganisms and are divided into three classes (Figures 1 and 2). Class 1 consists of stable and identified strains of microorganisms propagated by companies. Class 2 includes selected microorganisms, isolated and then propagated by companies. Finally, Class 3 includes microorganisms propagated using traditional methods.
 Figure 1: Products tested with different compositions and origins A-876
Figure 1: Products tested with different compositions and origins A-876
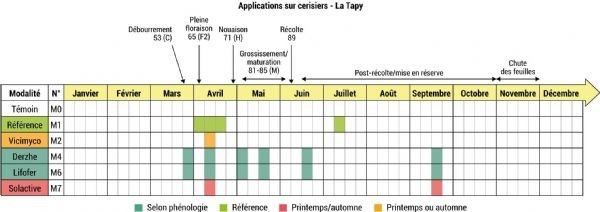 Figure 2: Application scheme according to protocol A-876 STDI
Figure 2: Application scheme according to protocol A-876 STDI
The trial includes a negative control plot. This plot should not receive any fertiliser for the entire duration of the study. However, an exception was made for the GiSelA® 6 rootstock present in the experimental plot. Indeed, the complete absence of fertiliser would lead to tree death. This dwarfing rootstock has a poorly developed root system, making it highly susceptible to errors in management practices. The plot also has a history of marked deficiencies, with chlorosis appearing rapidly in the event of insufficient fertiliser input during the growing season. As a result, fertiliser applications were reduced for this treatment. The fertilisation programme for the control treatment is detailed in Figure 3.
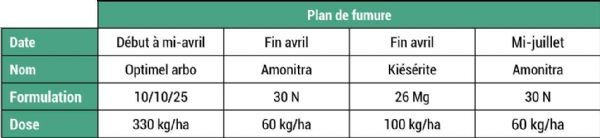 Figure 3: Fertilisation programme - typical applications during the trial period (reference) A-876
Figure 3: Fertilisation programme - typical applications during the trial period (reference) A-876
The study is conducted on cherry trees of the Summit variety, a mid-season variety widely grown in orchards in the Vaucluse region. The extensive orchard was planted in 2019. The trees are trained in a vase shape and irrigated using a drip irrigation system. The six treatments are randomly distributed within the orchard. Each treatment consists of three consecutive trees and is repeated three times. Measurements are taken on the central tree to avoid any edge effects. The aim of the trial is to establish a correlation between the tree’s physiological response and the development of soil biomass following the application of biofertilisers. Several types of information are collected and analysed (Figure 4):
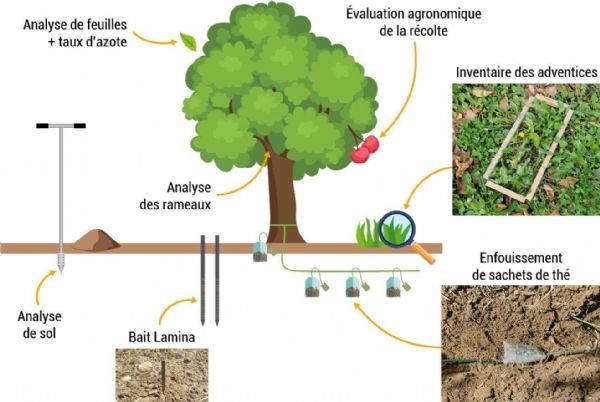 Figure 4: The technical indicators observed during production are varied A-876 STDI
Figure 4: The technical indicators observed during production are varied A-876 STDI
Soil indicators: what impact did the tested solutions have?
A first line of investigation aims to assess the impact of biofertilisers applied to the soil. To monitor the amount of nitrogen available in the soil, regular samples were taken from all treatments. The purpose of these data is to provide information on available quantities and reveal how these quantities vary over time. The results vary considerably from year to year. In 2022, for example, nitrate levels decreased in May in the Lifofer treatment. This suggests that this biostimulant may be retained in the soil for a shorter period than the others and may require more frequent applications. However, this trend was not observed in 2023 (Figure 5) or in 2024, when levels increased at the second reading for this treatment. Surprisingly, nitrate levels are sometimes lower in the reference plot than in the less fertilised control, suggesting significant heterogeneity within the plot.
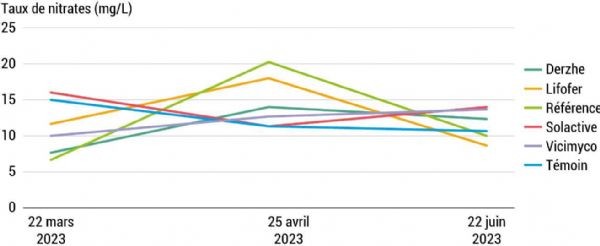 Figure 5: Nitrate levels differed only slightly between treatments following A-876 STDI
Figure 5: Nitrate levels differed only slightly between treatments following A-876 STDI
Contrary to expectations, the treatments with Vicimyco and Solactive did not show lower nitrate levels, despite not being nitrogen-based. In fact, the Vicimyco treatment contains, in addition to endomycorrhizal fungi, brown seaweed extracts, which are rich in calcium, potassium, sodium or chloride (Dhargalkar & Pereira, 2015). The Solactive product is a consortium of nitrogen-fixing bacteria and inorganic phosphorus-solubilising bacteria. However, if the added bacteria are inactive due to soil conditions, these biostimulants provide little benefit. Dherze treatments contain 1.3% organic nitrogen, while Lifofer treatments contain forest litter. These compositions may explain the nitrate levels, which are sometimes higher than in the control. At the end of the season, all treatments converged at lower nitrogen levels, reflecting plant uptake. Overall, nitrogen levels in the plot are low, which may have created a favourable environment for the introduction of biostimulants and the resulting production of differences. However, these values do not show significant differences between the various measurement dates.
The presence of microorganisms in the soil is essential. However, these microorganisms must also be able to effectively decompose any added element. To assess this capacity, one analysis involves measuring the decomposition rate of labile organic matter using green tea bags. Labile organic matter corresponds to active organic matter and ensures short-term soil fertility. Two-gram bags of previously dehydrated tea were buried at the base of the central tree in each plot, at a depth of around ten centimetres. The objective was to characterise soil life by monitoring their decomposition 90 days after burial. A second bag was left exposed on each tree. Analysis of the residual weights of these exposed bags revealed no significant differences between treatments. This confirms that conditions are similar across the plot and provides a reference value for weight loss in bags when they are not buried.
The weight of the buried bags is shown in Figure 6. Weight loss was greater for the buried bags, ranging from 41% to 45% of their mass, compared with those left in the open air, which lost between 23% and 36% of their mass. As expected, given the variability of these data, no statistically significant difference was observed between the different treatments.
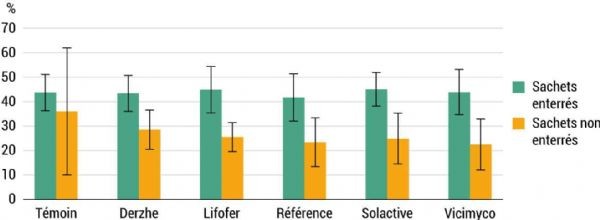 Figure 6: Average change in tea bag weight during the three years of the trial A-876 STDI.
Figure 6: Average change in tea bag weight during the three years of the trial A-876 STDI.
To complement this analysis of soil life, a second variable was observed. The Bait Lamina test makes it possible to monitor biotic processes in the soil by tracking the degradation of the substrate by fauna, particularly invertebrates and, to a lesser extent, the activity of microorganisms and fungi. This test consists of placing plastic strips with 16 holes filled with a substrate composed of 70% cellulose and 30% various substrates (eucalyptus residues, beans or wheat, or a commercial substrate) that can be consumed by soil fauna. A correlation can be established between the number of holes consumed and the biological activity of the soil. Indeed, the greater the number of holes consumed, the higher the biological activity of the soil. However, these measurements did not reveal any significant differences between the treatments observed.
Finally, attention focused on weeds. In fact, the flora present in a plot reflects the quality and characteristics of its soil. One-square-metre quadrats were used to count the plant species present within them. Certain species indicate compaction, significant erosion and waterlogging, which can lead to a slowdown in microbial life or even anaerobic conditions (Veronica persica, Taraxacum sp., Picris hierachioides) (Ducerf, 2014). Conversely, other species associated with an excess of organic matter and nitrogen had disappeared by 2024 (Bromus sterilis, Bromus ramosus, Cirsium arvense, Muscari sp., Ornithogalis sp., Hedera helix, etc.). The species and their abundance did not vary between the treatment methods studied.
The second part of this study focused on the trees’ ability to store nutrients for the following year. At the end of winter, 30 one-year-old shoots were collected from each treatment group and sent to an external laboratory for analysis. The results of these analyses did not reveal any significant differences. However, these measurements remain a reliable indicator of the trees’ ability to build up their reserves at the end of the season. They provide information on their vegetative status during the previous year. Nitrogen levels were adequate and well balanced with calcium, indicating good plant health. After a 2022 season marked by high temperatures, nitrogen levels were imbalanced in relation to calcium. This type of imbalance often indicates a water-related issue. Similarly, potassium levels were adequate, but the balance was too weak in relation to calcium, a symptom of limited water availability. These observations were not repeated in 2023 and 2024, which were certainly hot summers, but less extreme than 2022. Finally, carbohydrate levels were correct and therefore sufficient to allow the trees to begin growing in spring.
Leaf analyses, carried out during the summer according to a similar protocol, confirmed these observations but did not allow us to distinguish between the different treatments tested.
Numerous observations were made during harvest. Average yields, detailed in Figure 7, varied considerably from year to year. In 2022, the trees had a high fruit load, with an average of 17.4 t/ha across the entire plot, but the fruit was small in size (41% measured 26–28 mm). In the following two years, yields were closer to expected values: 8.4 t/ha in 2023 and 6.2 t/ha in 2024. These values are low compared with the average for cherry orchards, which stands at between 10 and 12 t/ha. The trees in this plot are planted too extensively (low density per hectare) for the dwarfing rootstock used, which explains these results.
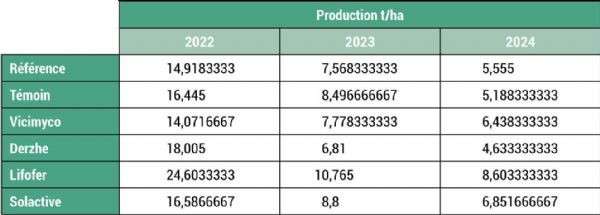 Figure 7: Yield variations between treatments for each monitoring year A-876
Figure 7: Yield variations between treatments for each monitoring year A-876
Production values vary depending on the treatment. A trend appears to emerge: the Lifofer treatment shows higher production rates across all monitoring years. However, these results must be interpreted with caution because, due to the significant variability of the data, it is impossible to conclude statistically that there was an increase in average yield. Furthermore, given the high heterogeneity of tree growth within the plot, it is important to rule out the possibility that the Lifofer treatment was applied to more vigorous and therefore more productive trees. In this case, trunk cross-sectional area is measured annually during winter, and production is expressed as a proportion of trunk cross-sectional area, as shown in Figure 8.
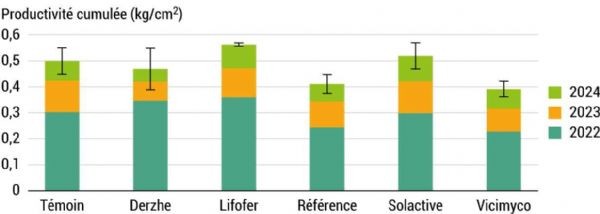 Figure 8: Relationship between tree production and vigour A-876 STDI
Figure 8: Relationship between tree production and vigour A-876 STDI
For some treatments, the production ratio is reversed. The Lifofer treatment still shows a slight increase in production associated with lower yield variability. However, here again, the statistical analysis does not allow us to conclude that the products used had an influence on production in the experimental plot.
Fruit quality was also observed at harvest. No statistically significant differences were found in fruit size distribution (Figure 9), colour, firmness, sugar content or acidity.
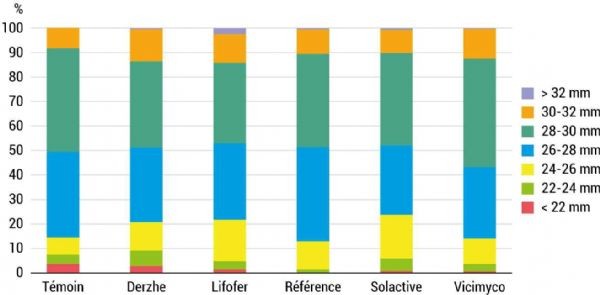 Figure 9: The treatments did not influence the average fruit size of A-876 STDI
Figure 9: The treatments did not influence the average fruit size of A-876 STDI
The analyses show no impact on the quality of tree production. However, it is possible that the trees assimilate more nitrogen depending on the conditions. To test this hypothesis, leaf nitrogen levels are measured during the fruit ripening period and again a few days after harvest. An N-tester® is used: this clip-shaped instrument allows rapid diagnosis of nitrogen nutrition in a growing crop. These measurements follow the same trend every year. Figure 10 presents the results for 2022, as this was the year in which the greatest number of measurements was taken. The readings show a gradual increase in leaf nitrogen levels until 14 June. The average value therefore reached 566.17, compared with 411.05 at the start of the measurements. Subsequently, the average values remained stable. These trends correspond well to the expected development of the plant, which reaches a stable phase during the summer. However, no significant difference was detected between the six trial treatments on any of the recorded dates, regardless of the year of measurement.
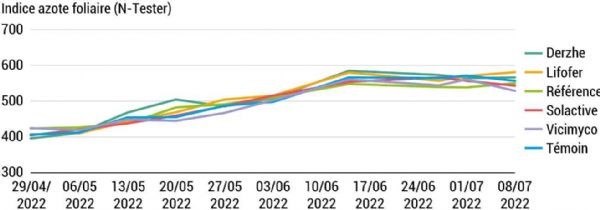 Figure 10: Evolution of leaf nitrogen content A-876 STDI
Figure 10: Evolution of leaf nitrogen content A-876 STDI
Several observations allow us to assess tree vigour. An evaluation carried out during the summer provides information on stress symptoms. Those related to climatic stress, such as yellow spots on the foliage or curled leaves, were observed in 2023, but to a lesser extent than in 2022. Although the heat was intense, it was shorter-lived than in 2022; the trees therefore withstood the summer better. The same was true in 2024. The observations were identical for yellow and curled leaves. Overall, these symptoms appeared in all individual plots, with a similar incidence depending on the treatment method. The treatments do not appear to have had any impact on the general condition of the trees.
This project examines a wide range of variables to assess the effect of the preparations tested on both soil microflora and plants. Although no tangible effect of the products emerged across all the variables studied during these three years, this may be due to the mechanism of action of these substances, which may require time to induce a measurable effect. Therefore, a multi-site analysis over a longer period will be even more important in order to draw conclusions on the potential effectiveness of the products tested.
Unlike soil application, most studies on seaweed-extract-based biostimulants applied to cherry trees focus on the effects of foliar application. In the Sweetheart and Skeena varieties grafted onto GiSelA® 6, an extract of Ascophyllum nodosum (also used in Vicimyco) reduces cracking rates and increases fruit width, weight, size, pH and wax content, but has no effect on nutritional characteristics or fruit load (Correira et al., 2015). When combined with salicylic acid and soy betaine, these seaweed extracts help increase total soluble solids, pH, polyphenols, vitamin C and antioxidant compounds in the Staccato variety (Goncalves et al., 2020). They also improve fruit size, colour and, consequently, ripening uniformity.
However, the use of biostimulants is not intended to completely replace conventional practices, but rather to complement them. For example, irrigation remains necessary in cherry cultivation in the Mediterranean region, but this practice increases soil salt content, which affects nitrification (Sindhu et al., 1967; Mac Clung et al., 1987).
Nevertheless, some studies have shown that the application of biostimulants, whether microbial or otherwise, improves nutrient-use efficiency and tolerance to abiotic stresses such as drought, extreme temperatures or salinity (Calvo et al., 2014; Bulgari et al., 2015; Haplern et al., 2015; Povero et al., 2016; Yakhin et al., 2017). They could therefore be among the possible solutions to support the agricultural transition in the face of climate change. For the benefits of biostimulants to become tangible, farming practices must evolve towards a more rational use of plant protection products, for example for weed control or the management of diseases and pests, and move towards more sustainable solutions for soil health.
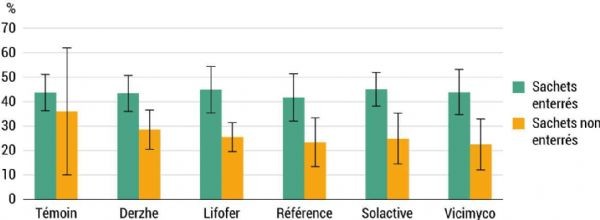 Figure 11: Average change in tea bag weight during the three years of the trial A-876 STDI.
Figure 11: Average change in tea bag weight during the three years of the trial A-876 STDI.
Key points
Do biostimulants support fruit production in trees? - Three-year review of an evaluation project on cherries
At CTIFL’s “La Tapy” experimental station, an experimental programme is underway to assess the impact of biofertilisers on cherry cultivation, with the aim of improving soil fertility and the sustainability of practices. Several microorganism-based products were tested on Summit cherry trees grafted onto GiSelA® 6 to observe their effects on the soil (nitrates, biological activity) and on the plant (growth, yield, resilience, quality). After three years, no significant differences were found between treatments. The Lifofer product shows a slight trend towards higher yields, but there is no formal evidence of its effectiveness. Biostimulants appear promising, but their effectiveness depends strongly on the initial soil conditions.
Aliénor ROYER-LANOOTE, CTIFL
Martin PHAM, CTIFL
Source: CTIFL NEWS 410 – January 2026
Opening image source: Stefano Lugli